Glide, Part II: phase diagram & glide values
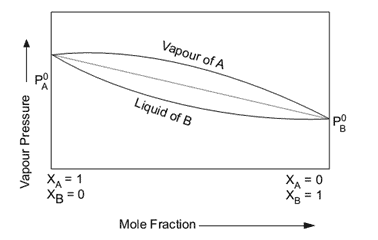
In fluid mixtures, azeotropic formation is the exception rather than the rule: the bubble and dew curves, always considered in the simplifying example of two-component mixtures, therefore take on the characteristic appearance of the “lens” phase diagram shown in the figure:
The transformations of phase change at constant pressure evolve accordingly at a variable temperature (glyde temperature) in the range between the dew temperature and the bubble temperature corresponding to the composition of the mixture. During an evaporation process, for example, the most volatile component (low boiling point) first separates from the liquid phase of the mixture, which is progressively enriched, as a consequence, by the higher boiling component. The opposite happens during the condensation process.
During the phase change, the composition of the liquid phase and the vapour phase are therefore different from each other (fractionation): at a given temperature the different composition values of the liquid phase and the vapour phase can be measured on the bubble and dew curves respectively at the intersection points with the corresponding isotherm (horizontal line).
The following figure illustrates how, at constant pressure, the temperature of phase change varies in the bi-phase equilibrium region, giving origin to the phenomenon of “glide” of evaporation and condensing temperatures along the heat transfer surfaces. In a practical way, lower saturation temperature values are measured at the evaporator inlet than at the outlet, and vice versa, higher saturation temperature values are measured at the condenser inlet rather than at the heat exchanger outlet.
In practice, as long as a zeotropic mixture has glide values lower than 2°K, its use in refrigeration circuits does not pose significant problems compared to the use of “homologous” one-component refrigerating fluids or in an azeotropic mixture (as in the case of R410A, for example), to the point that it is sometimes referred to as quasi-azeotropic mixtures. On the other hand, when the temperature glide is higher than the mentioned value, it is no longer allowed to ignore this circumstance in the design of the refrigerating circuit and in the operations connected to it.
Francesco Viola, Eng.